It’s Time to Heal is a special package about engineering the future of bio and healthcare. See more at: https://a16z.com/time-to-heal/.
In biotech, platforms — the combination of technology and scientific expertise — are the product engines that generate novel drug candidates. One important measure of a platform’s value is how many “shots on goal” — promising drug development programs — it can generate. The rise of highly-productive, industrialized platforms are poised to reshape the biotech industry with their potential to pursue novel targets or generate multiple drugs through iteration on a central technology.
Let’s take the company of the moment, Moderna, as an example. Over the last 10 years, the company has built a machine to produce mRNA-based therapeutics at incredible speed and scale, generating a wide range of drug development programs spanning infectious disease, immuno-oncology, and rare disease. In the earliest days of the COVID-19 pandemic, they were able to apply their platform to design a successful mRNA vaccine against SARS-CoV-2 in just 48 hours. It’s a testament to the power of their platform that they were able to jump on an emerging disease while continuing to advance their broad pipeline.
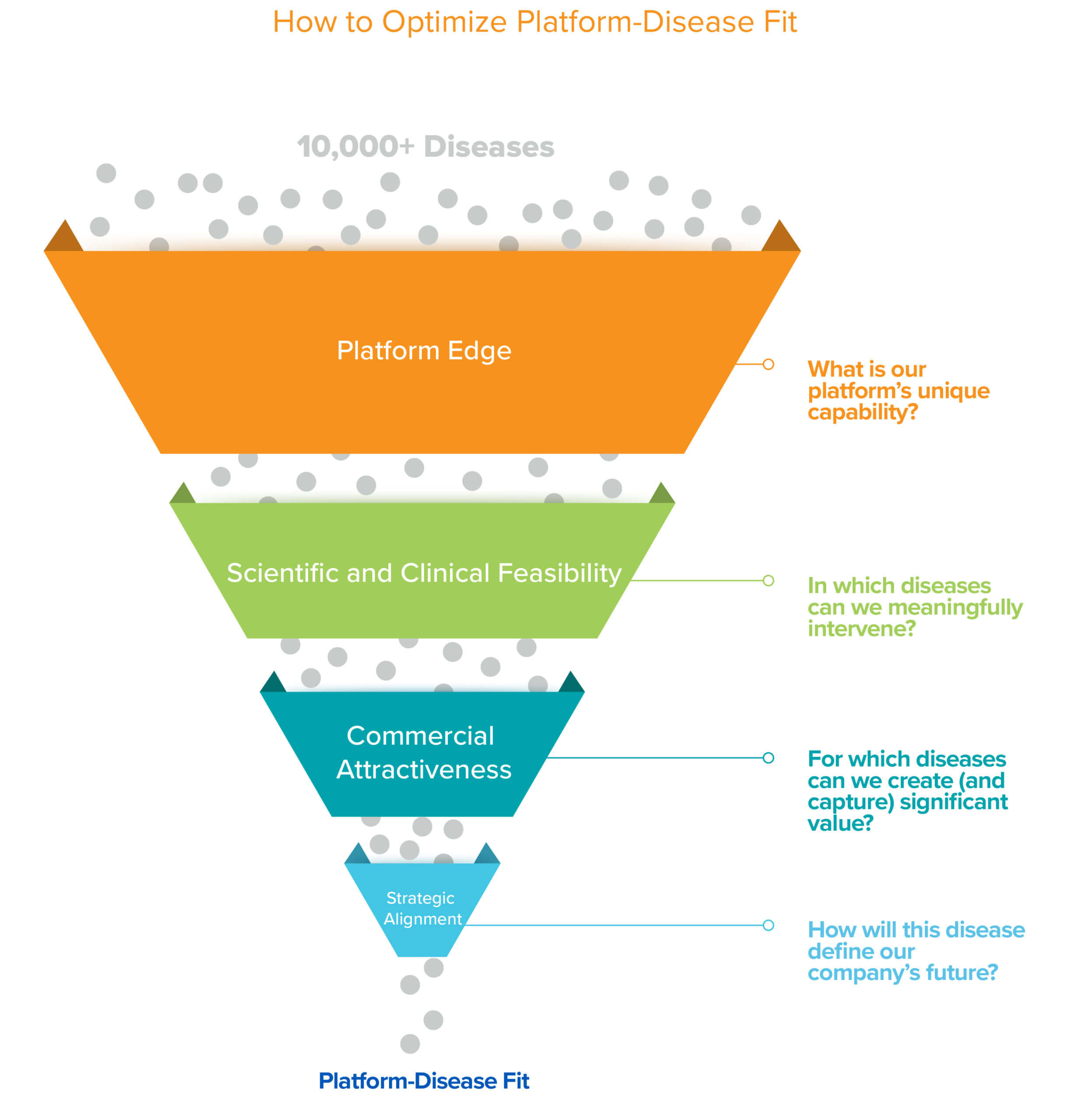
Platforms, by their very nature, have the potential to solve many problems. But for startups with promising platforms, yet limited resources, it’s important to call your shot. In the earliest days of company-building, founders are rightly focused on building teams and technology, which can understandably delay them prioritizing and choosing which specific diseases to pursue. Biotech is a tough business; finding the best possible match between the platform and disease, or platform-disease fit (PDF), is essential to success. The only way to truly demonstrate the worth of a platform, and thus a platform company, is to optimize for PDF early on to deliver a valuable solution to a real-world problem. Given that there are on the order of 10,000+ known human diseases and every platform and company is unique, finding that perfect PDF is no simple task. While it is a puzzle with no single objectively correct solution, we provide a framework in this piece that we hope is helpful for thinking through this critical decision.
Platform edge
Platforms come in many flavors, from programmable medicines (e.g. cell and gene therapies) that can theoretically target a broad universe of disease targets, to clever new approaches or modalities (e.g. bispecifics or PROTACs) that can hit elusive or “undruggable” targets, to platforms that can help us discover novel targets for complex diseases. Optimizing PDF should start from a first principles analysis of what novel superpower your platform provides that others can’t access. It’s not enough to solve a problem; what problem can you uniquely solve? The platform and science should guide you.
Optimizing Platform-Disease Fit should start from a first principles analysis of what novel superpower your platform provides that others can’t access. It’s not enough to solve a problem; what problem can you uniquely solve? The platform and…For instance, if you have a new gene delivery platform that can carry >5 kb cargo or a new nuclease that can do >10kb insertions, a natural place to start is looking at diseases that could be treated with such large-scale gene replacements. On the other hand, if you have an editing platform that can elegantly perform single base changes with minimal off-targets, SNP based diseases should be the focus.
Similarly, there are a number of highly validated and valuable disease targets that were historically difficult to drug with conventional modalities like small molecules or biologics. Take for example KRAS, one of the most well characterized and causative oncogenes. It was deemed an undruggable holy grail, but new platforms have recently uncovered how to hit it, inspiring a fierce competition within and between big pharma companies and startups. If your platform has an advantage in accessing these high-value targets, this can be a rich vein to mine. You should let your weapon guide you to the battles where you have an advantage.
Another way to think about your platform and its superpower is whether it is better suited for generating new drugs or identifying new targets for drugs. The latter case is incredibly useful for many complex diseases, like many metabolic diseases, that suffer from a dearth of validated targets and treatment options. If you have a platform that has an unbiased ability to uncover novel targets, you should go after the diseases where there is a deep unmet need for new therapeutic strategies.
In sum, for this initial step of identifying platform edge, the more specific you can get, the better. Now, if you get to this step and you don’t have a strong case for your platform’s unique angle, STOP HERE. An undifferentiated platform will not survive in such a competitive and capital-rich environment as we currently have. Take this as a sign that your company idea may not be ready for prime time; work on refining or refocusing your platform. The reality is that not all new technologies translate into great platform companies capable of developing novel medicines (and that’s okay, as it could still be an invaluable tool for science).
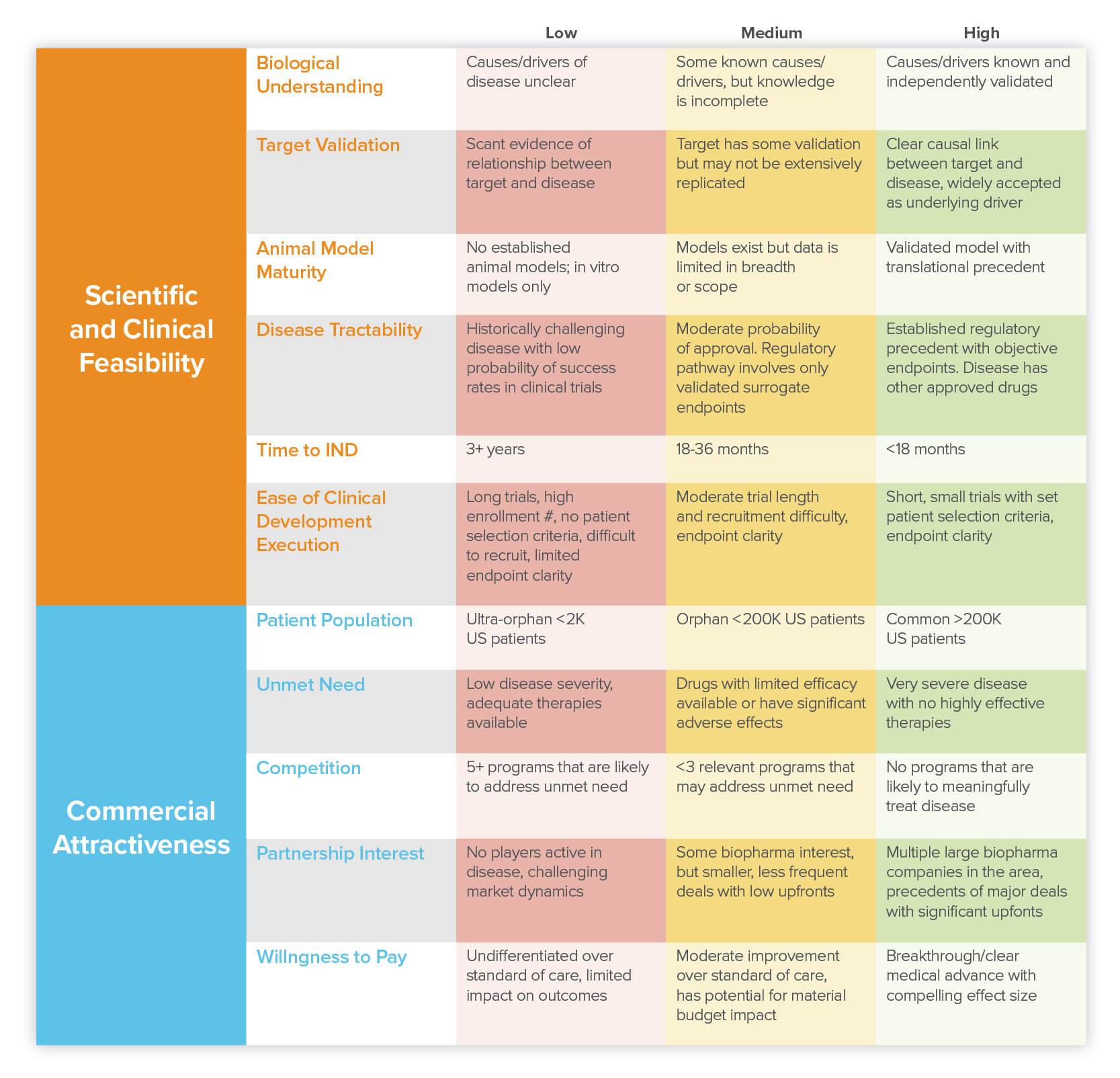
For those who have identified their superpower and have begun to narrow down a list of diseases that play to their strengths, the next steps are even less formulaic and there is likely a spectrum of answers on a sliding scale depending on your goal. It is important to think through two core axes: scientific/clinical feasibility and commercial attractiveness. Within each of these axes there are several key factors to consider and for each, we can assign a low, mid, or high scoring that maps to how best to think about prioritization for a particular factor (the higher the better).
Scientific and Clinical Feasibility
Determining scientific and clinical feasibility is all about thinking through the diseases where your platform has the potential to most effectively help patients. The idea here is to hone in on diseases where you believe you can meaningfully intervene and improve patient outcomes. Usually that means having a strong understanding of both the biology and clinical aspects of the disease.
On the scientific side, it’s important that you have (or can develop) a solid theory of what drives that disease and how your platform-based intervention would act. In an ideal situation, you would know what is causing the disease, have data showing that modulating a particular target in humans leads to improvements, have handy biomarkers that can be used to validate that success, and have an established animal model for screening. But of course, this is biology, so cases where all the checkboxes are so clearly defined are rare. So what do your PDF candidates have? In the table above, we break down the key components to consider when assessing scientific and clinical feasibility.
If the goal of your platform company is to pursue novel interventions or new modalities for well-characterized diseases, it is best initially to go after diseases that tick the most “high” boxes. Let’s take CAR-T cells, for example. Achieving scientific feasibility for this modality looks like finding diseases where, ideally, (1) there is a specific cell type or types that directly drive the disease, (2) it’s known that destroying those cells improves the disease state, (3) it’s known that destroying those cells (or any downstream effects) would not harm the patient and (4) those disease-causing cells express a surface protein that is not expressed on normal cells which can be used as the CAR target.
But if your platform is best suited for target discovery, this advice can be partially flipped on its head. Diseases where there is limited understanding of the disease drivers, or specifically a dearth of novel targets, are ideal opportunities for these platforms. So target discovery platforms can consider diseases that score lower on some of the items in the scientific feasibility table (above), but importantly would need to be able to demonstrate that their target discovery platform does indeed identify valuable targets — an important aspect of determining PDF is being able to demonstrate this fit on a reasonable timeline.
So you should consider time to POC (proof-of-concept), especially for novel platforms and modalities. For some diseases it is much easier and faster to demonstrate that a particular approach is sound or that an intervention is effective. In infectious diseases, the results from animal models closely correlate with those from humans, meaning that you can get a meaningful readout of the success of your therapy without doing highly-regulated human studies — in other words, you can generate high confidence before starting clinical trials. To take an extreme example, contrast this with therapies that target aging, where delayed aging or changes in lifespan could take a literal lifetime to prove. Which is why many aging companies focus on targeting the effects of age-related diseases versus aging itself. In diseases where animal models don’t exist or do not predictably or accurately replicate human disease, POC may not be achievable until a signal has been generated in a human clinical trial — a milestone which can take many years and millions of dollars to reach. So picking one disease over the other could dramatically impact time to initial POC of your platform, and in turn, time to ultimately validate your business.
So picking one disease over the other could dramatically impact time to initial proof-of-concept of your platform, and in turn, time to ultimately validate your business.An equally important measure is your platform’s productivity, and for therapeutics companies, that’s measured in the ability to generate IND (investigational new drug) applications. These filings with the FDA are the starting gun for clinical programs. “Time to IND” has historically been used by investors as a gauge to determine when a company can raise its next round of capital, including possibly tapping the public markets via an IPO. It’s also an important factor for potential partners. While some business development (BD) deals are done solely around a platform’s technology potential, many partners seek to fill or augment their early-stage pipeline, thus programs that are at or near IND become attractive ways to “slot in” innovation into dry pipelines.
If INDs are the starting gun, clinical trials can be both marathon and obstacle course. Evaluating the terrain by determining the clinical feasibility of testing your drug candidate must also be carefully assessed. How relatively easy or difficult it is to run a clinical trial depends on a number of variables. Aspects like how clear the specific endpoints are, how many patients you would need to enroll, how big the patient population is, and how long the trial needs to run can all have a big effect. And perhaps most importantly from a practical perspective: do you have strong patient selection criteria (like a biomarker) that can help you rapidly identify and recruit the right patients? This is an especially important consideration in heterogeneous diseases (where you want to recruit only the subset of patients that are most likely to benefit from your therapy) or diseases where there are many other drug candidates competing to enroll patients for their trials too.
But you don’t always need large patient cohorts. Though it may seem really difficult to find and recruit patients for a super rare disease trial, if your new allele specific gene editing system has really large effect sizes, you would only need to enroll a small number of patients to reach sufficient statistical power. On the flip side, you might have technology that can modulate a common neuromuscular disease, but because the particular biology of the disease is highly complex and multifaceted with lots of confounding effects, the trial may take much longer to run and require far more patients to achieve a statistically significant result.
Minimizing clinical risk is especially important when dealing with novel platforms and modalities. There is a reason why the first genome editing indications were sickle cell anemia and blindness. For these diseases, you can administer the gene therapy ex vivo or in the contained environment of the eye, respectively, which is much less risky than systemic administration. In the former case, ex vivo editing allowed the researchers to check their work and do rigorous QA on the cells before administering back into the patient, greatly increasing the safety of this new technology. Clever initial choices like these can afford you more leeway while still allowing you to demonstrate the features that really make your platform shine.
There are, of course, some diseases that are just tough to crack. The probability of success for a historically challenging multi-dimensional disease like Alzheimer’s is fundamentally different from that of a rare monogenic disease with clear genetic drivers. Does that mean that no one should ever try and make new therapies for Alzheimer’s? Of course not (and this is prime ground for a target discovery platform company), but it is up to you to decide how clinically feasible it is for your platform and perform your own risk/reward calculation.
Commercial Attractiveness
At this stage, you should have a list of diseases that your platform is uniquely qualified to tackle and that have strong scientific and clinical feasibility. Now it’s time to think about creating value and commercial interest. The key here is to identify diseases where your platform-derived therapeutic has the potential to either address an unmet need, significantly improve upon the standard of care, or substantively impact the cost of care.
The key here is to identify diseases where your platform-derived therapeutic has the potential to either address an unmet need, significantly improve upon the standard of care, or substantively impact the cost of care.Start by thinking about your potential patient demographic and market size. It goes without saying that, in the ideal case, you’d make a drug for an important disease (or multiple diseases, in the case of some blockbusters) that can be widely administered to a large patient population. A quintessential example here is the monoclonal antibody-based drug Humira, a TNF alpha inhibitor. It treats a wide range of autoimmune conditions affecting millions of patients and generates on the order of $20B annual revenue.
But bigger is not necessarily always better. Some diseases, like certain neurodegenerative diseases, may impact a huge demographic but come with a history of late-stage failures. And don’t confuse patient population for market size either. Some diseases have small patient populations, like certain rare genetic diseases, but because a new platform technology may be able to cure them (as opposed to treat them), they can command high prices — Novartis’ gene therapy treatment Zolgensma for somatic muscular atrophy, a devastating and fatal disease in children, launched with a $2.1M price tag.
Another aspect to factor in is how desperately a new therapeutic option is needed by patients. The last thing you want is to do is develop a novel, and likely expensive, drug for a disease that is a relatively minor inconvenience to patients or that can be controlled or mitigated by alternative means. Even for important chronic diseases like cardiovascular disease, the bar is high for another biologic or a new experimental gene therapy given the proven safety and efficacy of statins. Even if it ends up showing differentiated improvement, can patients (and payors) afford it and would they be willing to pay a premium? Alternatively, if a disease has no other effective treatments and the current patient outcomes are poor, even a small patient population can be commercially very attractive, making the value proposition much easier to justify.
The commercial potential of a space is of course influenced by the broader competitive landscape. Who else is working on this disease and can possibly beat you to the market? Or worse, render your drug obsolete? How likely are they to succeed? Are they (or are you) protected by an IP moat? You need to have 360-degree vision. It’s not only commercialized products, but also pipeline programs within larger pharma or smaller biotechs. Watch out for icebergs; companies may have under-the-radar programs that only emerge once they’re fairly advanced. And given the explosion of novel modalities, there’s also increasing potential for competition from directly comparable modalities (e.g. CAR-T versus CAR-NK) as well as traditional modalities like small molecules or biologics. Oh, and don’t forget about substitutes — alternatives like surgery count too.
Lastly, sometimes the best signals on the commercial attractiveness come in the form of heat — the proverbial phrase you’ll hear from investors, founders and industry veterans alike: “How hot is this space?” Look for evidence of active BD or M&A interest from biopharma companies. Given the harsh realities of market dynamics, there are some areas, like infectious diseases or antibiotics (where even approved drugs can lead to bankrupt companies) where it’s been extremely difficult to land a deal in recent history (…but could that change in a post-pandemic world?). On the contrary, super hot areas like immuno-oncology or neurological diseases — where cell and gene therapies have the potential to be curative — can command immense terms like the $300M upfront ($2Bn total deal size) Adaptive Biotech deal with Genentech or the $175M upfront CRISPR Therapeutics deal with Vertex, just to name a few recent ones. Even though the core goal of this exercise is to find your prized PDF for development within your own company, strong external industry interest in a particular area can bring huge tailwinds to your program.
Strategic Alignment
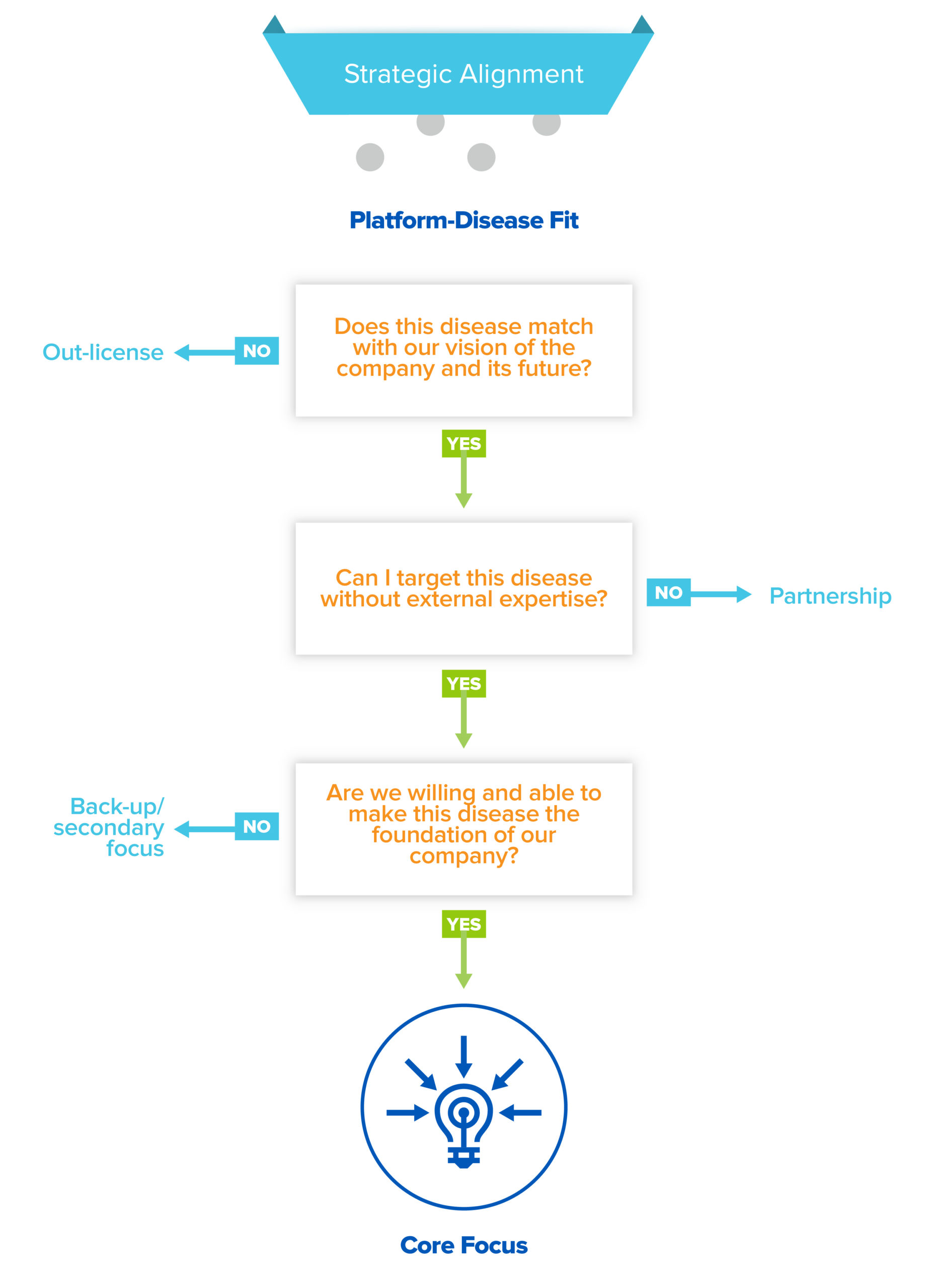
So what should the focus within your company be? After meticulously whittling down your list to the diseases that pass the platform edge, scientific/clinical feasibility, and commercial attractiveness filters, you have finally found your PDF picks! But while you may love them equally, you still have to pick your favorite child, as there are only so many programs a single company can work on. To do that, you need to assess opportunities using a last, critical filter: strategic alignment. One way to think of this: PDF tells you what you can work on, but this will tell you what you should work on.
Your pool of PDF candidates have three fates: out-license, partner, or core internal focus. Perhaps you are not personally motivated to develop a program in cosmetic medicine, despite high clinical feasibility and commercial attractiveness. In that case, you should out-license (read: sell) the rights to use your platform to develop assets in that disease area. Out-licensing not only forces focus, it’s also a good way to capture value for otherwise latent assets.
In other cases, a particular disease may be a compelling choice, but requires extensive external expertise or infrastructure in order to succeed. In that case, a partnership may be in the cards. BD partnerships are the lifeblood of the industry, allowing innovators and incumbents to co-create and share value. But don’t just focus on economics; partnerships can be incredibly valuable for startups because they enable you to learn and develop capabilities not only on your partner’s dime but also under their tutelage. For early stage companies, a partner with a world-class franchise in a competitive disease area, like immuno-oncology, can provide access to translational or clinical expertise that would otherwise take years to build. Or they could provide access to complex, expensive infrastructure, like cell therapy manufacturing, that you wouldn’t be able to build yourself.
But hopefully, there will also be goldilocks opportunities in your shortlist, where you are uniquely positioned to drive towards meaningful value creation, where you have the potential to go-it-alone and build the necessary capabilities in-house to be successful. And if you have more than one or two opportunities, others can be held in reserve for when you are ready to expand the scope of your platform and company. Keep in mind all of the downstream implications of choosing a disease as a core focus; it impacts who you hire and how you build certain functions, like your R&D competencies or clinical operations. As you grow, you’re growing roots — you can quickly find yourself from “working on liver disease” to becoming a “liver disease” company.
Finding your PDF is an art and not a science, even though we hope to give you structured canvas to begin sketching out your journey. Choose wisely, as you are drawing the destiny of your company.
Acknowledgements: Thank you to Camp4 Therapeutics for sharing their thoughts and framework for indication selection, many of which have been adapted for this piece.