“Using AI to design a fleet of AAV delivery vehicles to create the FedEx for gene therapy.”
Gene therapy, with its potential to alter genes inside a patient’s cells, could be one of our most powerful tools in medicine—it holds the promise of “one-and-done” cures for genetic diseases. When it comes to these diseases, we often know precisely the genetic payloads we want to deliver, but don’t always have the right vehicles to deliver them into the relevant cells in the body. Adeno-associated virus (AAV), considered to be largely harmless to humans, is the primary workhorse vehicle (vector) we use today to deliver therapeutic genes in the body. But AAV has its limitations: it may not deliver enough of its genetic payload to the right cells, it’s difficult to manufacture, it can have serious safety issues at very high doses, and many patients are not candidates for AAV-based therapies because they have pre-existing immunity to certain AAVs. We currently have just two approved gene therapies, but the industry hopes to have as many as 10-20 cell and gene therapy approvals per year by the end of 2025. Gene therapy has the potential to treat—and even cure—thousands of devastating diseases. Can AAVs deliver?
AAVs’ therapeutic potential was originally observed over 20 years ago. Since then, scientists have pursued multiple approaches to discover, evolve, or design novel AAVs with enhanced properties and characteristics. Can we find an AAV that specifically targets, say, the central nervous system in order to treat neurodegenerative diseases? Or design an AAV that’s easier to manufacture? It’s a hard mountain to climb. AAV capsids (shells) are delicate, complex 20-sided structures. When it comes to manipulating them in order to improve them, one false step can result in a precipitous “fall” that breaks the capsid. And even if you can make progress on optimizing one property (like tissue-specific targeting), it often comes at the significant expense of others (like manufacturability and immunogenicity). Which is why traditional approaches—directed evolution or rational design, or even the hope of discovering other naturally occurring AAVs—have rarely yielded improved capsids. A more powerful approach is needed to escape the limitations of natural AAVs and fully realize the promise of gene therapy.
Dyno’s Ascent
In rock climbing, a “dyno” is a rapid move across a rock face in order to reach the next hold. Eric Kelsic, who studied physics at Caltech and trained at George Church’s famed Harvard lab, was thinking about the toughest, most valuable problems artificial intelligence (AI) could tackle in biology—and realized that AI was an ideal tool to accelerate the search for better AAVs. In 2018, he teamed up with machine learning expert (and fellow Church Lab alum) Sam Sinai, Adrian Veres, Tomas Bjorklund, seasoned biotech entrepreneur Alan Crane, and George Church to found Dyno Therapeutics. Dyno’s mission is to create AI-designed AAV vectors with transformative delivery properties to cover the vast landscape of human diseases. The company’s CapsidMap platform comprehensively surveys AAV topography and finds the best routes to optimize multiple parameters in parallel in order to rapidly generate more efficient, highly specific, and less immunogenic AAVs. In comparison to traditional methods, Dyno’s AI-driven approach has already proven to be highly prolific: they have designed enhanced liver-targeting variants of naturally occurring AAV capsids and screened billions of potential sequences in silico using machine learning models to design thousands of viable synthetic AAV capsids—validating the use of advanced machine learning models to design highly diverse, functional AAV capsids.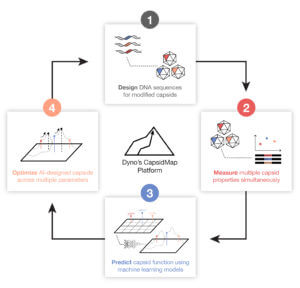 In addition to creating transformative therapies, productive bio platforms can enable novel business models too. If AI is an ideal tool for designing a diverse array of novel AAVs with varied functions, AI-designed AAVs can accelerate and reshape the entire gene therapy industry. From its inception, Dyno chose to eschew the traditional approach of investing in its own product pipeline (i.e. developing its own gene therapies) and instead decided to build the platform that will serve as the horizontal “infrastructure” layer to supply novel AAVs to power all gene therapies. Dyno’s approach has already proven to be highly prolific here too: the company has signed partnerships, potentially worth over $4 billion in aggregate, to develop next-generation AAV vectors for gene therapy leaders like Sarepta in muscle diseases, Novartis in eye diseases, and Roche/Spark Therapeutics for the central nervous system and liver. And this is just the start of their climb.
In addition to creating transformative therapies, productive bio platforms can enable novel business models too. If AI is an ideal tool for designing a diverse array of novel AAVs with varied functions, AI-designed AAVs can accelerate and reshape the entire gene therapy industry. From its inception, Dyno chose to eschew the traditional approach of investing in its own product pipeline (i.e. developing its own gene therapies) and instead decided to build the platform that will serve as the horizontal “infrastructure” layer to supply novel AAVs to power all gene therapies. Dyno’s approach has already proven to be highly prolific here too: the company has signed partnerships, potentially worth over $4 billion in aggregate, to develop next-generation AAV vectors for gene therapy leaders like Sarepta in muscle diseases, Novartis in eye diseases, and Roche/Spark Therapeutics for the central nervous system and liver. And this is just the start of their climb.
I am thrilled to announce that a16z is leading the Series A investment in Dyno. This capital will help expand their team and dramatically accelerate the design and development of improved AAV vectors targeting areas currently inaccessible to gene therapies—like lung, heart, and kidney diseases. I am honored to join the Board of Directors and to partner with CEO Eric Kelsic and team as Dyno delivers on its vision to scale new heights in gene therapy.
* * *

- Novartis CEO Vasant Narasimhan on Transforming a 250-Year-Old Company Jorge Conde and Vasant Narasimhan
- Investing in Phylo Jorge Conde and Zak Doric
- Operating on DNA is more like surgery than medicine Jorge Conde
- Wartime vs Peacetime: Ben Horowitz on Leadership Ben Horowitz and Jorge Conde
- Can America Win The AI Biotech Race Against China? | Lada Nuzhna & Elliot Hershberg Lada Nuzhna, Elliot Hershberg, Jorge Conde, and Erik Torenberg
